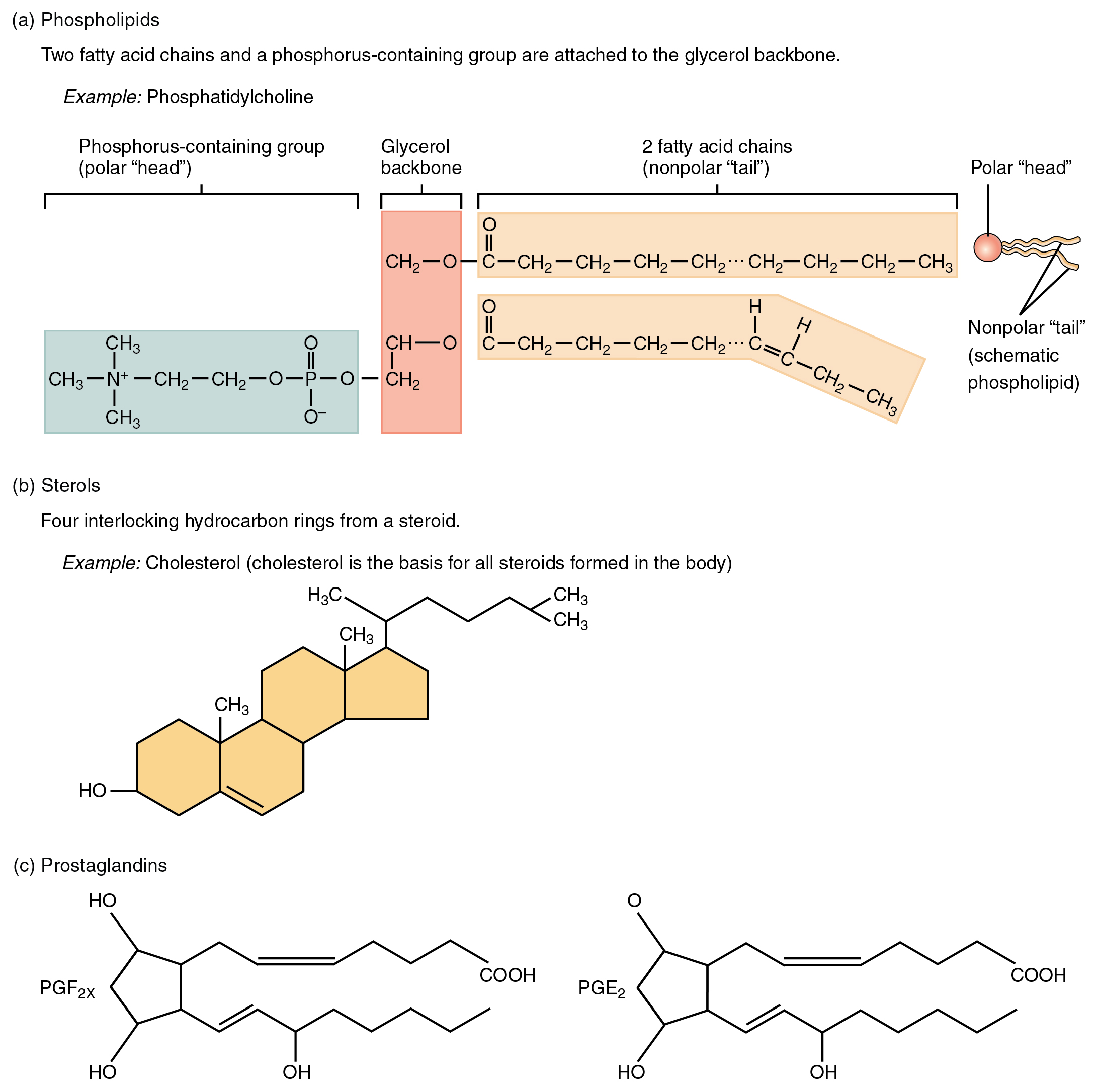
Derivatives of PGE 2 are now used in the United States to induce labor. Their wide range of physiological activity has led to the synthesis of hundreds of prostaglandins and their analogs. Aspirin and other nonsteroidal anti-inflammatory agents, such as ibuprofen, obstruct the synthesis of prostaglandins by inhibiting cyclooxygenase, the enzyme needed for the initial step in the conversion of arachidonic acid to prostaglandins. Slight structural differences give them highly distinct biological effects however, all prostaglandins exhibit some ability to induce smooth muscle contraction, lower blood pressure, and contribute to the inflammatory response. The prostaglandins are among the most potent biological substances known. Subscripts are attached at the end of these abbreviations to denote the number of double bonds outside the five-carbon ring in a given prostaglandin. The five major classes of prostaglandins are designated as PGA, PGB, PGE, PGF, and PGI. It is now known that they are synthesized in nearly all mammalian tissues and affect almost all organs in the body. They are called prostaglandins because they were originally isolated from semen found in the prostate gland. They are unsaturated fatty acids containing 20 carbon atoms and are synthesized from arachidonic acid-a polyunsaturated fatty acid-when needed by a particular cell. Prostaglandins are chemical messengers synthesized in the cells in which their physiological activity is expressed. \): Lipid Organization Based on Structural Relationships
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
